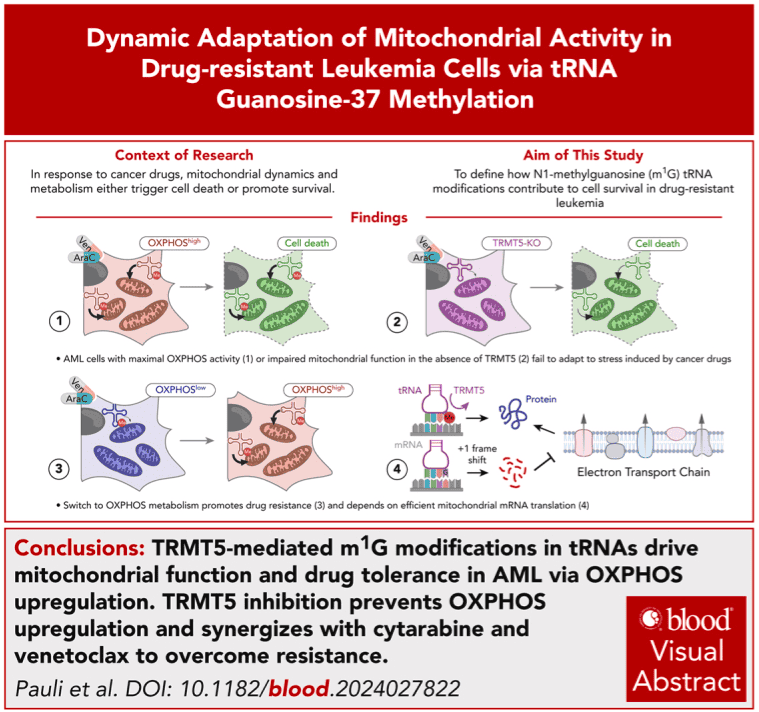
Disrupting tRNA modifications to target mitochondrial vulnerabilities in drug-resistant leukemia cells
Authors
Key Points
- TRMT5-mediated N1-methylguanosine tRNA modifications drive mitochondrial function and drug tolerance in AML via oxidative phosphorylation.
- TRMT5 inhibition prevents oxidative phosphorylation upregulation and synergizes with cytarabine and Ven to overcome resistance.
Published
2025